
Fibrinet® PRF Wound Matrix is designed to support the body’s natural healing processes by delivering a structured, autologous fibrin matrix directly to the wound site. The result is a simple, point-of-care solution that integrates seamlessly into advanced wound management protocols while leveraging the patient’s own biologic potential.
Autologous PRF Matrix For Wound Management
Key Features of the Fibrinet® PRF Wound Matrix System
- The system provides intact, concentrated platelets in a platelet-rich fibrin membrane graft without thrombin2,5
- Sustained availability of platelets and associated growth factors out to seven days; ability to proliferate cells in vitro2-4
- Easy-to-use, closed system with the highest platelet capture efficiency and reproducibility5
- Under the supervision of a healthcare professional, the PRP gel produced may be topically applied for the management of cutaneous wounds, including: Diabetic ulcers, Venous leg ulcers, Mechanically or surgically debrided wounds.
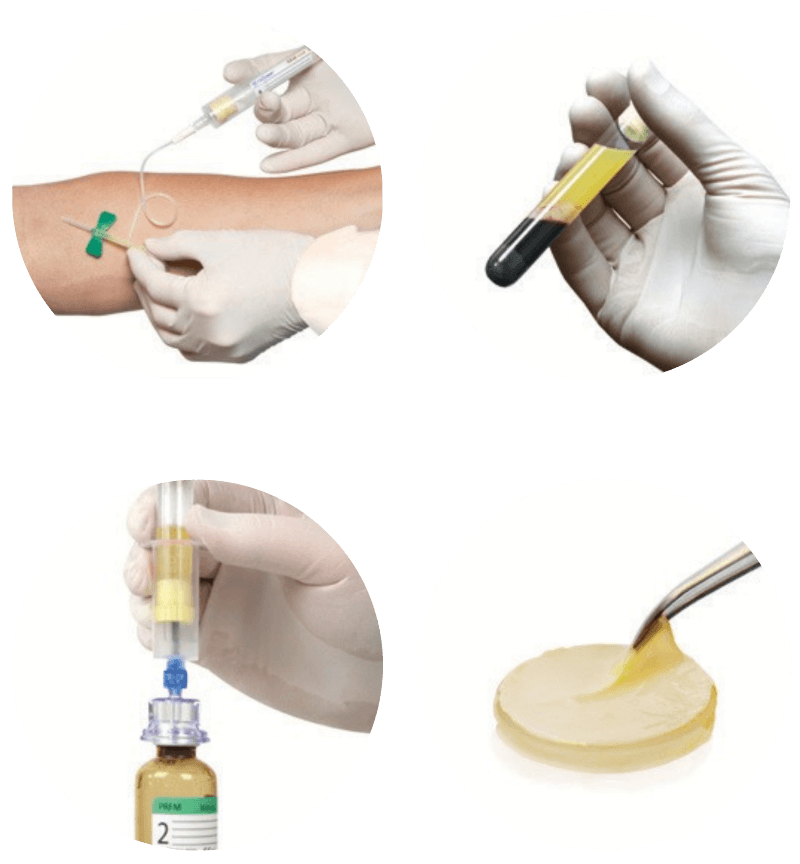
How It Works

Coverage
- Single Kit: Covers up to 6 cm2
- Double Kit: Covers up to 12 cm2

Structual Strength

Biological Activity
Simple, Safe Point-of-care Preparation
Draw
Spin
Transfer

Spin

Apply
Core Elements of Tissue Repair
Scaffold
The resulting PRFM demonstrates enhanced mechanical properties compared with conventional fibrin clots, with stiffness up to 600× greater and mechanical characteristics comparable to arterial tissue.2
Signals
- VEGF – associated with angiogenesis and the formation of new blood vessel formation2,8
- PDGF-A/B – involved in cellular recruitment and matrix formation during tissue repair2
- TGF-B1 – involved in regulation of fibroblast activity during tissue repair2
Cells
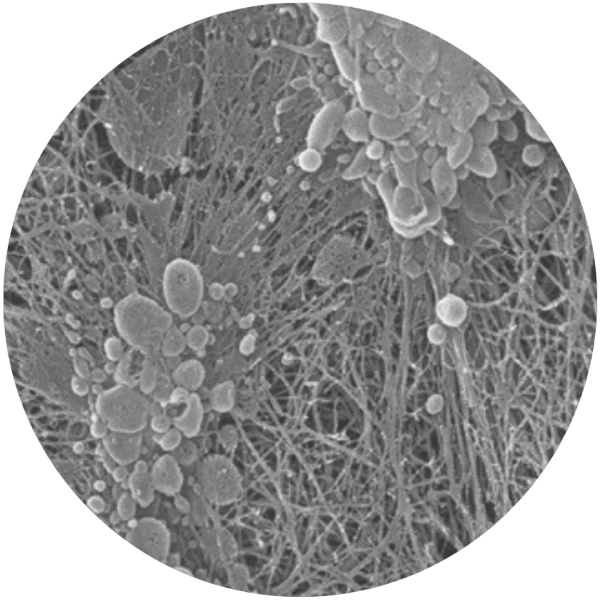
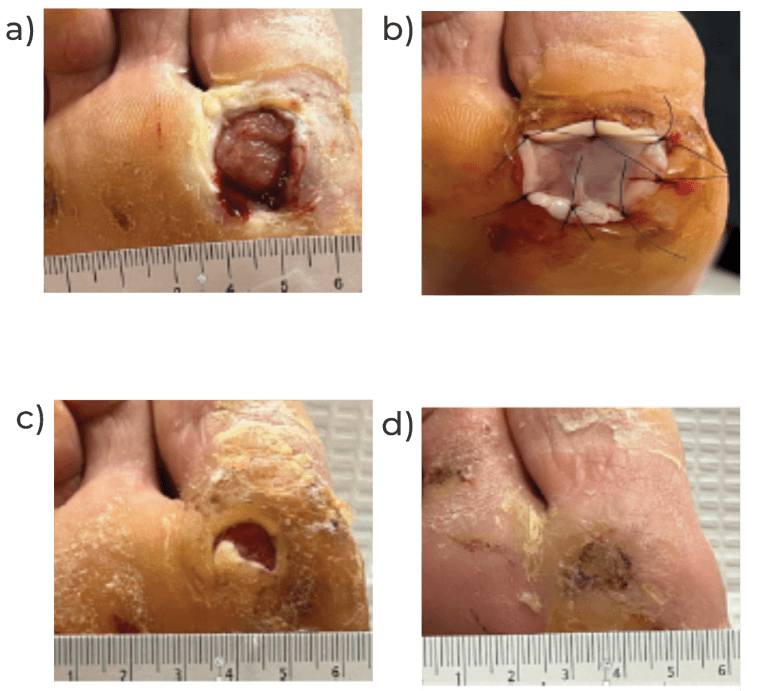
Image 1: 65 year old male with history of ulcer submetatarsal right #1 for 6 months a) Screening Visit [SV1] b) Application Visit, 2 weeks post-screening [TV1], c) Treatment Visit #3 [TV3], d) Ulcer closed on Treatment Visit #5 [TV5] 1
Reimbursement
Fibrinet® PRF Wound Matrix is 510k approved for wounds. Royal Biologics is committed to physicians and facilities using Fibrinet® PRF Wound Matrix to benefit their patients. Learn more about the coding, billing, and reimbursement below:
G0465 – Diabetic Chronic Wounds
$2,107.97
$83.84
$1,064.49**
($920.19-$1,509.27)
20 Weekly Applications
2
G0460 – Non-Diabetic Chronic Wounds
Carrier Priced. Coverage determined by MAC;
Not Covered per NCD
1
MUE
National Coverage
Our proprietary, patented technology supports the use of advanced biologic products that may be eligible for coverage under Medicare and certain Medicare Advantage plans.
Effective for services performed on or after April 13, 2021, the Centers for Medicare & Medicaid Services (CMS) will cover autologous PRP for the treatment of chronic non-healing diabetic wounds under section 1862(a)(1)(A) of the Social Security Act (the Act) for a duration of 20 weeks, when prepared by devices whose Food and Drug Administration-cleared indications include the management of exuding cutaneous wounds, such as diabetic ulcers.
- 20 weekly applications
- Chronic Diabetic Wounds (head to toe)
- Not subject to skin substitutes documentation and policy limitations
G0465 – Diabetic Chronic Wounds , FDA-cleared device
Autologous platelet rich plasma (PRP) or other blood-derived product for diabetic chronic wounds/ulcers, using an fda-cleared device for this indication, (includes as applicable administration, dressings, phlebotomy, centrifugation or mixing, and all other preparatory procedures, per treatment)
G0460 – Non-Diabetic Chronic Wounds
Autologous platelet rich plasma (PRP) or other blood-derived product for NON-diabetic chronic wounds/ulcers, using an fda-cleared device for this indication, (includes as applicable administration, dressings, phlebotomy, centrifugation or mixing, and all other preparatory procedures, per treatment)
- ONE Diabetic Diagnosis code (E Code)
AND - ONE Non-Pressure Chronic Ulcer Code (L Code)
| E08 | E09 | E10 | E11 | E13 | |
|---|---|---|---|---|---|
| Location | Diabetes Due to Underlying Condition | Diabetes Drug or Chemical Induced | Diabetes Type I | Diabetes Type II | Diabetes Other |
| Foot | E08.621 | E09.621 | E10.621 | E11.621 | E13.621 |
| Other | E08.622 | E09.622 | E10.622 | E11.622 | E13.622 |
| Location | Breakdown of Skin | Fat Layer Exposed | Necrosis of Muscle | Necrosis of Bone | Muscle Involvement w/o Evidence of Necrosis | Bone Involvement w/o Evidence of Necrosis | Other Specified Severity |
|---|---|---|---|---|---|---|---|
| Lower Extremity | |||||||
| Left | |||||||
| Thigh Left | L97.121 | L97.122 | L97.123 | L97.124 | L97.125 | L97.126 | L97.128 |
| Calf Left | L97.221 | L97.222 | L97.223 | L97.224 | L97.225 | L97.226 | L97.228 |
| Ankle Left | L97.321 | L97.322 | L97.323 | L97.324 | L97.325 | L97.326 | L97.328 |
| Heel & Midfoot Left | L97.421 | L97.422 | L97.423 | L97.424 | L97.425 | L97.426 | L97.428 |
| Other Parts of Foot Left | L97.521 | L97.522 | L97.523 | L97.524 | L97.525 | L97.526 | L97.528 |
| Other Parts of Leg Left | L97.821 | L97.822 | L97.823 | L97.824 | L97.825 | L97.826 | L97.828 |
| Right | |||||||
| Thigh Right | L97.111 | L97.112 | L97.113 | L97.114 | L97.115 | L97.116 | L97.118 |
| Calf Right | L97.211 | L97.212 | L97.213 | L97.214 | L97.215 | L97.216 | L97.218 |
| Ankle Right | L97.311 | L97.312 | L97.313 | L97.314 | L97.315 | L97.316 | L97.318 |
| Heel & Midfoot Right | L97.411 | L97.412 | L97.413 | L97.414 | L97.415 | L97.416 | L97.418 |
| Other Parts of Foot Right | L97.511 | L97.512 | L97.513 | L97.514 | L97.515 | L97.516 | L97.518 |
| Other Parts of Leg Right | L97.811 | L97.812 | L97.813 | L97.814 | L97.815 | L97.816 | L97.818 |
| Upper Extremity | |||||||
| Buttocks | L98.411 | L98.412 | L98.413 | L98.414 | L98.415 | L98.416 | L98.418 |
| Back | L98.421 | L98.422 | L98.423 | L98.424 | L98.425 | L98.426 | L98.426 |
| Abdomen | L98.431 | L98.432 | L98.433 | L98.434 | L98.435 | L98.436 | L98.438 |
| Chest | L98.441 | L98.442 | L98.443 | L98.444 | L98.445 | L98.446 | L98.448 |
| Neck | L98.451 | L98.452 | L98.453 | L98.454 | L98.455 | L98.456 | L98.458 |
| Face | L98.461 | L98.462 | L98.463 | L98.464 | L98.465 | L98.466 | L98.468 |
| Groin | L98.471 | L98.472 | L98.473 | L98.474 | L98.475 | L98.476 | L98.478 |
| Skin of Other Sites | L98.491 | L98.492 | L98.493 | L98.494 | L98.495 | L98.496 | L98.498 |
| Left | |||||||
| Upper Arm Left | L98.A121 | L98.A122 | L98.A123 | L98.A124 | L98.A125 | L98.A126 | L98.A128 |
| Forearm Left | L98.A221 | L98.A222 | L98.A223 | L98.A224 | L98.A225 | L98.A226 | L98.A228 |
| Hand Left | L98.A321 | L98.A322 | L98.A323 | L98.A324 | L98.A325 | L98.A326 | L98.A328 |
| Right | |||||||
| Upper Arm Right | L98.A111 | L98.A112 | L98.A113 | L98.A114 | L98.A115 | L98.A116 | L98.A118 |
| Forearm Right | L98.A211 | L98.A212 | L98.A213 | L98.A214 | L98.A215 | L98.A216 | L98.A218 |
| Hand Right | L98.A311 | L98.A312 | L98.A313 | L98.A314 | L98.A315 | L98.A316 | L98.A318 |
NCD 270.3 coverage applies to the following site of service:
• POS 11 – Office
• POS 12 – Home/private residence
• POS 19 – Off-campus hospital outpatient
• POS 22 – On-campus hospital outpatient
• POS 31 – Skilled Nursing
• POS 49 – Independent clinic
- Diabetes Diagnosis Code (see covered codes on chart per NCD)
- Chronic Wound Diagnosis Code (see covered codes on chart per NCD)
- Duration of Wound
- Baseline Measurements
- Conservative Care Measures Taken
- Measurements Post Conservative Care
The above documentation should be carried through to each PRP application and chart note
- PRP Treatment Application number
- Name of PRP system used
- Wound Measurements
- Treatment plan and patient progression
ORDERING INFORMATION
- Part NumberDescription
- PRF-01 Fibrinet PRF Wound Matrix, Single Kit
- PRF-02 Fibrinet PRF Wound Matrix, Double Kit
REFERENCES
1. Fridman R, Wielgomas J. A Prospective Study to Evaluate Efficacy and Safety of Autologous Platelet-Rich Fibrin Matrix for the Treatment of Chronic Diabetic Foot Ulcers. Clin Res Foot Ankle. 2024;12:590. doi:10.4172/2329-910X.1000590.
2. Lucarelli E, et al. A recently developed bifacial platelet-rich fibrin matrix. Eur Cells Materials 2010; 20:13-23.
3. Roy S, Sen CK, et al. Platelet-rich fibrin matrix improves wound angiogenesis via inducing endothelial cell proliferation. Wound Rep Regen. 2011;19(6):753-766.
4. Visser LC, Arnoczky SP, Caballero O, et al. Platelet-rich fibrin constructs elute higher concentrations of TGF-ß1 and increase tendon cell proliferation over time when compared to blood clots of similar volume: A comparative in vitro analysis. Vet Surg. 2010c; 39(7):811-817.
5. Castillo TN, Pouliot MA, Kim HJ, Dragoo JL. Comparison of growth factor and platelet concentration from commercial PRP separa- tion systems. Am J Sports Med. ePub Nov 4, 2010.
6. Roy S., Driggs J., Elgharably H., et al. Platelet-rich fibrin matrix improves wound angiogenesis via inducing endothelial cell proliferation. Wound Repair Regen. 2011;19:753-766.
7. O’Connell SM, Impeduglia T, Hessler K, Wang X-J, Carroll RJ, Dardik H.Autologous platelet-rich fibrin matrix as cell therapy in the healing of chronic lower-extremity ulcers. Wound Repair and Regeneration. 2008;16(6):749–756. doi:10.1111/j.1524-475X.2008.00426.x
8. Johnson KE, Wilgus TA. Vascular Endothelial Growth Factor and Angiogenesis in the Regulation of Cutaneous Wound Repair. Adv Wound Care (New Rochelle). 2014 Oct 1;3(10):647-661. doi: 10.1089/wound.2013.0517. PMID: 25302139; PMCID: PMC4183920.
Explore our Entire Product Line
Autologous Advanced Biologics
Advanced Synthetics
Wound Care
About Royal
Royal is a pioneering leader in orthobiologic and wound care research and development. Royal’s commitment lies in delivering top-tier, innovative biologic solutions that enhance the healing process and improve patients' quality of life. Royal places patient well-being at the center of its mission, offering exceptional solutions to address a variety of complex needs.
At Royal, our mission is driven by patient-centered care, innovation and excellence, and integrity and compassion. Royal provides a comprehensive array of products tailored to meet the varied needs of the orthobiologic and wound care industry.
As a rapidly expanding force in the industry, Royal is continuously developing and launching new products and solutions that set benchmarks for quality, effectiveness, and patient outcomes. Royal’s commitment to research and innovation keeps it at the forefront of the industry, helping to shape the future of healthcare and improving lives.
