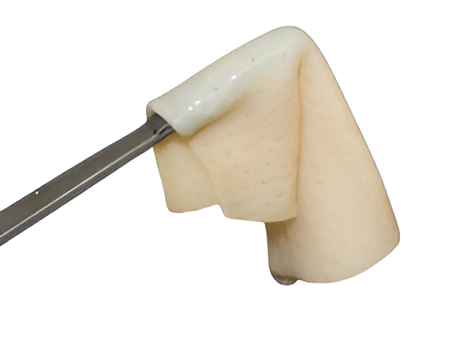
Preservation of the Extracellular Matrix (ECM)
- CoriLuxeDL is produced using proprietary methods that avoid harsh chemicals, which could compromise the microstructure by weakening the collagen network or cross-linking the tissue. The matrix component and structure of CoriLuxeDL were evaluated against unprocessed skin tissue and CoriLuxe using histology, immunohistochemistry, mechanical, and ELISA to assess the preservation of the extracellular matrix. It demonstrated that CoriLuxeDL is similar to that of native tissue in terms of the structure and bioactive components.
- The basement membrane is physically removed, and the porous natural structure is preserved after processing. Surgeons do not need to identify the orientation of CoriLuxeDL.
- Growth factor screening assay detected significant levels of bFGF, EGF-R, HGF, IGF, IGFBPs, PDGF, TGFa, TGFb1 & VEGF in CoriLuxeDL.
- Glycosaminoglycans (GAGs) and Angiogenin were detected in CoriLuxe.
- The tensile test shows that the deep cut layer is slightly less strong than the top layer from the same donor. However, testing across three donors revealed no statistically significant difference. Proprietary sterilization with low-dose precision ebeam irradiation to achieve a Sterility Assurance Level (SAL) of 10-6.
- CoriLuxeDL is to be used for the repair or replacement of damaged or inadequate integumental tissue or for other homologous uses of human integument.
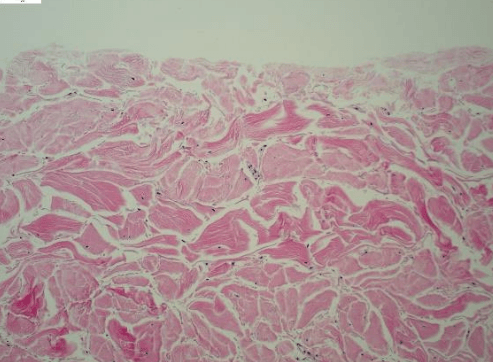
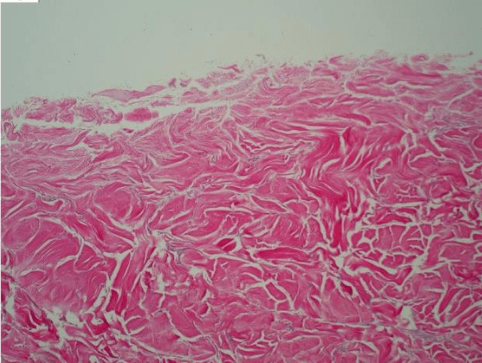
Fig. 1
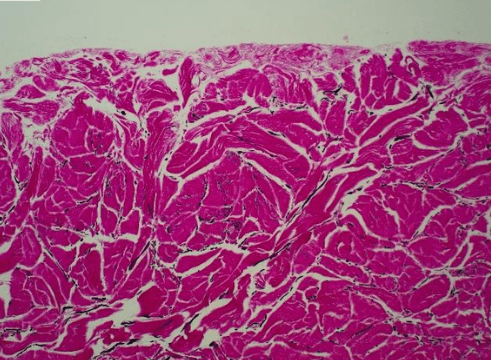
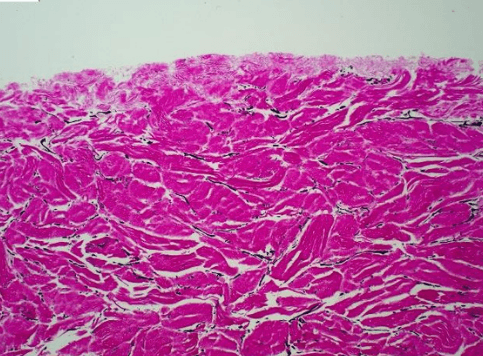
Fig. 2
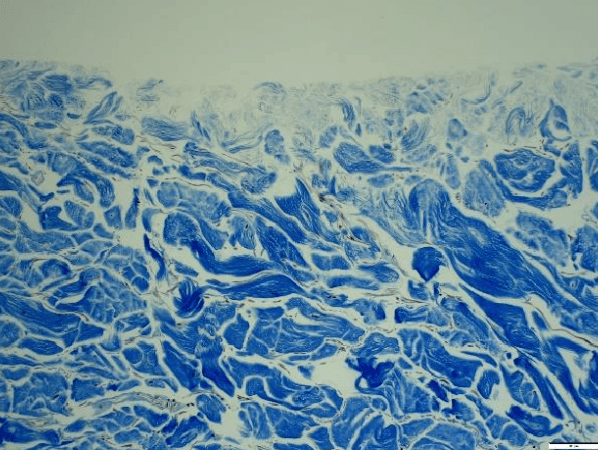
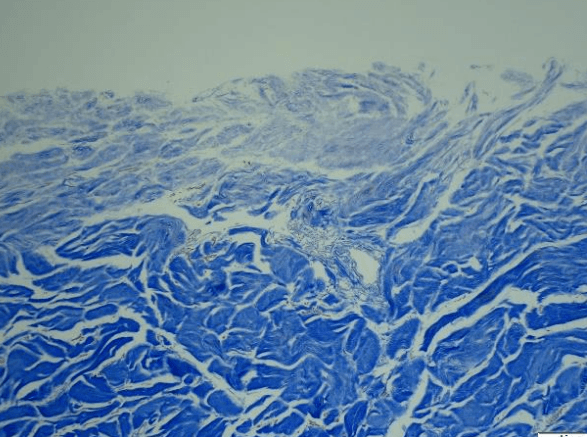
Fig. 3
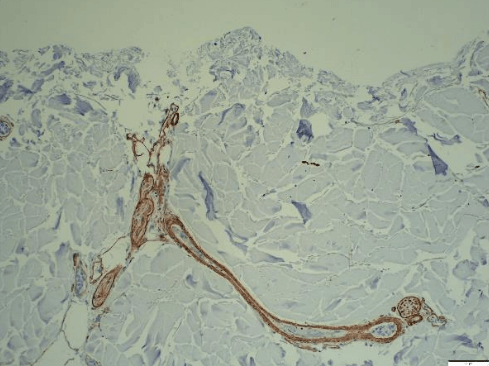
Fig. 4
Product Ordering Information
- Code Description
- CLD0404 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 4×4 cm
- CLD0407 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 4×7 cm
- CLD0412 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 4×12 cm
- CLD0510 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 5×10 cm
- CLD0612 CoriLuxe DL Deep-layer Hydrated Acellular Dermal Matrix 6×12 cm
- CLD0812 CoriLuxe DL Deep-layer Hydrated Acellular Dermal Matrix 8×12 cm
- CLD1010 CoriLuxe DL Deep-layer Hydrated Acellular Dermal Matrix 10×10 cm
- CLD1212 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 12×12 cm
- CLD1616 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 16×16 cm
- CLD1620 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 16×20 cm
- CLD2020 CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix 20×20 cm
- CLD0404-TN CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix Thin 4x4cm
- CLD0510-TN CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix Thin 5x10cm
- CLD1012-TN CoriLuxeDL Deep-layer Hydrated Acellular Dermal Matrix Thin 10x12cm
- CLDM0404 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 4x4cm
- CLDM0407 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 4x7cm
- CLDM0510 CoriLuxeDL Deep-Layer Meshed Hydrated Acellular Dermal Matrix 5x10cm
- CLDM1212 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 12x12cm
- CLDM1018 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 10x18cm
- CLDM1616 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 16x16cm
- CLDM1620 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 16x20cm
- CLDM2020 CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix 20x20cm
- CLDM0404-TN CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix Thin 4x4cm
- CLDM0510-TN CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix Thin 5x10cm
- CLDM1012-TN CoriLuxeDL Deep-layer Meshed Hydrated Acellular Dermal Matrix Thin 10x12cm
References
- Badylak, S. F., et al. Extracellular matrix as a biological scaffold material: structure and function. Acta biomaterialia, 5(1), 1-13.
- Badylak, S. F. et al. The extracellular matrix as a scaffold for tissue reconstruction. In Seminars in cell & developmental biology (Vol. 13, No. 5, pp. 377-383). Academic Press.
- Chung, L. et al. Key players in the immune response to biomaterial scaffolds for regenerative medicine. Advanced drug delivery reviews, 114, 184-192. 4:
- J. Hur, et al. Outcome Assessment According to the Thickness and Direction of the Acellular Dermal Matrix after Implant-Based Breast Reconstruction. Biomed Res Int. 2021 Nov 16;
- Londono, R. et al. Biologic scaffolds for regenerative medicine: mechanisms of in vivo remodeling. Annals of biomedical engineering, 43(3), 577-592.
- Salbach, J., et al. Regenerative potential of glycosaminoglycans for skin and bone. Journal of molecular medicine, 90(6), 625-635. 8:
- WY Han, et al. Acellular Dermal Matrix without Basement Membrane in Immediate Prepectoral Breast Reconstruction: A Randomized Controlled Trial. Plast Reconstr Surg . 2024 Oct 1;154(4):649e-655e.
- Wen, J., et al. Applications of differential scanning calorimetry for thermal stability analysis of proteins: qualification of DSC. Journal of pharmaceutical sciences, 101(3), 955-964.
- Xiao SC, et al. The role of pores in acellular dermal matrix substitute. Ann Burns Fire Disasters. 2006;19(4):192-195
Explore our Entire Product Line
Autologous Advanced Biologics
Advanced Synthetics
About Royal
Royal is a pioneering leader in orthobiologic and wound care research and development. Royal’s commitment lies in delivering top-tier, innovative biologic solutions that enhance the healing process and improve patients' quality of life. Royal places patient well-being at the center of its mission, offering exceptional solutions to address a variety of complex needs.
At Royal, our mission is driven by patient-centered care, innovation and excellence, and integrity and compassion. Royal provides a comprehensive array of products tailored to meet the varied needs of the orthobiologic and wound care industry.
As a rapidly expanding force in the industry, Royal is continuously developing and launching new products and solutions that set benchmarks for quality, effectiveness, and patient outcomes. Royal’s commitment to research and innovation keeps it at the forefront of the industry, helping to shape the future of healthcare and improving lives.